- This page was created by volunteers like you!
- Help us make it even better. To learn more about contributing to MEpedia, click here.
- Join the movement
- Visit #MEAction to find support or take action. Donate today to help us improve and expand this project.
Vaccine
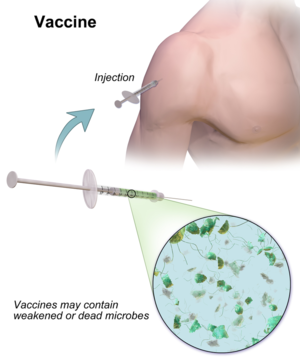
A vaccine is a biological preparation that provides active acquired immunity to a particular disease. A vaccine typically contains an agent that resembles a disease-causing microorganism and is often made from weakened or killed forms of the microbe, its toxins, or one of its surface proteins. The agent stimulates the body's immune system to recognize the agent as a threat, destroy it, and to further recognize and destroy any of the microorganisms associated with that agent that it may encounter in the future. Vaccines can be prophylactic (example: to prevent or ameliorate the effects of a future infection by a natural or "wild" pathogen), or therapeutic (e.g., vaccines against cancer are being investigated).[1][2][3][4]
Risk in developing ME/CFS
According to the 2018 version of The Clinician’s Vaccine Safety Resource Guide Vaccines: "Vaccines currently routinely recommended to the general population in the U.S. have not been shown to cause fibromyalgia or chronic fatigue syndrome (CFS)."[5]
A 2015 complete population study of data from 2009 to 2012 in Norway, following the country's mass vaccination during the 2009 influenza A (H1N1) pandemic, found that vaccination produced no increased risk of ME/CFS. By contrast, infection with flu more than doubled the risk of developing ME/CFS.[6]
A minority of people with ME/CFS have reported developing it after receiving a vaccination, but a causal relationship has not been shown.[7]
In one case, a 43-year-old man with no previous ill health developed CFS after having five vaccinations, all containing aluminium adjuvants, within the same month.[8] Based on this case, Exley et al. (2009) anticipated that the mass human papillomavirus (HPV) vaccination program then recently begun in the UK would carry related risks, as that vaccine also used an aluminium-based adjuvant. That was not borne out: a number of different studies concluded there is no evidence Cervarix,[9] or the later Gardasil[10][11][12][13] and Gardasil 9,[10] HPV vaccines lead to developing chronic fatigue syndrome.[14][15]
Risk in developing Gulf War Illness
Vaccinations have been proposed as an etiological factor in Gulf War Illness, especially the mass vaccinations given prior to military deployment.[16]
Vaccination in the presence of elevated cortisol levels can drive cytokine expression toward Th2 dominance.[17][18] Th2 dominance is seen in ME/CFS. [19][20]
Vaccines in ME/CFS and Fibromyalgia patients
Risk of contracting an infectious disease such as COVID-19, flu or other viruses and bacterial diseases depends on exposure to the infection and the strength of the person's immune system. People with a weakened immune system are known as immunocompromised or immunosuppressed, for example people with AIDS, diabetes or malnutrition, or people taking immunosuppressant medications.[21]
ME/CFS includes immune symptoms including a sore throat, swollen lymph nodes, and flu-like symptoms, but patients are generally not considered to be severely immunocompromised, and immunosuppressant medications are unlikely to be used by most ME/CFS patients, this means that ME/CFS patients are not significantly more likely to experience life-threatening complications from COVID-19, viral or other infections―however some patients are already very severely ill due to very severe ME.[22][23][7] This means most ME/CFS patients would not be considered to be at very high risk of serious COVID-19 complications unless they had other known risk factors such as obesity, older age or other illnesses.[22]
ME/CFS patients, viruses and vaccines
Many vaccines exist for common viruses, and viruses are generally known to increase the severity of ME/CFS in a proportion of patients, either temporarily or permanently; there is a known risk of deterioration after a virus although it is unclear exactly how many ME/CFS patients experience this.[7]
Vaccine effectiveness in ME/CFS patients
A study in 2012 assessed immune system responses to the flu vaccine in people with ME/CFS, and took biological samples that showed the immune systems of ME/CFS patients responded similarly to those of healthy people. The study indicated a similar antibody response in ME/CFS patients after the vaccine, and it was likely to be as effective in protecting ME/CFS patients against flu.[24] The study did not include a long-term follow-up to assess duration of vaccine effectiveness or overall effect on ME/CFS symptoms.[24]
Immunocompromised people and vaccines
People who are severely immunocompromised either by a particular disease or treatment are advised to get additional vaccines due to their suppressed immune system being unlikely to fight off infections.[25] The CDC states that only live and bacterial vaccines should be avoided by immunocompromised people (vaccines containing a reduced amount of a live virus or other infection).[26][25] While having ME/CFS does not make a person severely immunocompromised, some people with ME/CFS may have other known risk factors.[22] Immunocompromised people who receive a vaccine that is not live and not a bacterial vaccine will not be able to produce as many antibodies as a healthy person so the vaccine will not work as well, but the reduced protection should reduce the severity of the virus and improve survival rates.[26][25] Immunocompromised people who choose to have a vaccine may be given a specific type of vaccine more suited to their needs, for example a flu vaccine injection instead of the flu nasal spray vaccine (because the flu nasal spray is a live vaccine).[27]
Autoimmunity and ME/CFS
The unproven autoimmune hypothesis states that a subtype of ME involves autoimmunity, which is an overactive immune system rather than a weakened immune response.[28] Evidence for this theory is limited.
Types
Live vaccines
Live vaccines contain either weakened or killed pathogens which cannot cause disease. Different types include:
- Whole-pathogen vaccines, for example the Harvix Hepatitis A vaccine, these provide very good immunity for either a lifetime. or a very long period of time
- Subunit vaccines that just contain the antigens needed.[29]
Live vaccines may not be suitable for some people, for example those who have received an organ transparent, or those with a weakened immune system, or people with long-term conditions.[30]
Inactivated vaccines
These may contain all the pathogen, which has been killed or inactivated in a lab, for example by heat or another method, or just the antigens needed, for example the newer whooping cough (pertussis) which has a lower risk of fever or swelling at the injection site.[29] Protection is not as strong as with live vaccines, so several booster shots are usually needed.[30]
Toxoid vaccines
These contain chemically inactivated toxins, for example the diphtheria and tetanus vaccines that protect against those bacteria.[29]
Subunit, recombinant, polysaccharide and conjugate vaccines
These vaccines do not contain the whole virus or pathogen, they may contain just particular pieces, e.g. a protein, sugar or part of the shell of the pathogen. This creates a very strong targetted immune response.[30]
Recombinant vaccines have been around since the 1970s and use DNA technology to combine two or more sources, e.g., antigen in the hepatitis B vaccine is a hepatitis B virus protein produced by yeast cells into which the genetic code for the viral protein has been inserted into. These may use virus-like particles (VLP) from the shell of a virus to cause immunity without the risk of infection.[29]
Subunit, recombinant, polysaccharide and conjugate viruses are suitable for people with weakened immune systems, chronic illness or other long-term health conditions as well as the general population. Examples include the current Hib (haemophilus influenzae type b) vaccines, HPV vaccines, and hepatitis B vaccine.[30]
Several booster shots may be needed.[30]
New types of vaccines
The first COVID-19 vaccines to be approved in the United States, UK and European Union are the Pfizer-BioNTech and Moderna messenger RNA (mRNA) COVID-19 vaccines, mRNA vaccines do not contain live or weakened COVID-19, so these vaccines are suitable for severely immunocompromised people as well as people in the general population and cannot cause COVID-19.[31]
A number of other new types of vaccines are being developed. A vaccine to protect against multiple diseases caused by mosquitos is being developed to cause an immune response to mosquitos saliva rather than a particular virus or bacterium.[29]
Publications
- 2018, Do Vaccines Cause Fibromyalgia or Chronic Fatigue Syndrome (CFS)? in The Clinician’s Vaccine Safety Resource Guide[5]
See also
Learn more
- Vaccine Basics[32] - Health and Human Services
- Vaccine Types[29] - NIAID
- Coronavirus (COVID-19) vaccines[33] - World Health Organization - with a details of vaccines in development
- mRNA vaccines - CDC
- Vaccination of Adults with weakened immune systems - CDC
- Vaccine Storage and Handling[34] - CDC
- 10 Things to Know about US COVID-19 Vaccine Plans[35]
- Vaccines[36] Video short - Blausen.com
References
- ↑ Melief C, van Hall T, Arens R, Ossendorp F, van der Burg S (2015). "Therapeutic cancer vaccines". J Clin Invest. 125 (9): 3401–12. doi:10.1172/JCI80009. Archived from the original on June 29, 2017.
- ↑ Bol K, et al. (2016). "Prophylactic vaccines are potent activators of monocyte-derived dendritic cells and drive effective anti-tumor responses in melanoma patients at the cost of toxicity". Cancer Immunology, Immunotherapy. 65 (3): 327–39. doi:10.1007/s00262-016-1796-7.
- ↑ Brotherton, J. (2015). "HPV prophylactic vaccines: lessons learned from 10 years experience". Future Medicine. 10 (8): 999–1009. doi:10.2217/fvl.15.60.
- ↑ Frazer, I (2014). "Development and Implementation of Papillomavirus Prophylactic Vaccines". J. Immunol. 192 (9): 4007–11. doi:10.4049/jimmunol.1490012. Archived from the original on August 11, 2017.
- ↑ 5.0 5.1 Dudley, Matthew Z.; Salmon, Daniel A.; Halsey, Neal A.; Orenstein, Walter A.; Limaye, Rupali J.; O'Leary, Sean T.; Omer, Saad B. (2018). Do Vaccines Cause Fibromyalgia or Chronic Fatigue Syndrome (CFS)?. Cham: Springer International Publishing. pp. 241–244. doi:10.1007/978-3-319-94694-8_35. ISBN 9783319946931.
- ↑ Magnus, Per; Gunnes, Nina; Tveito, Kari; Bakken, Inger Johanne; Ghaderi, Sara; Stoltenberg, Camilla; Hornig, Mady; Lipkin, W. Ian; Trogstad, Lill (November 17, 2015). "Chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME) is associated with pandemic influenza infection, but not with an adjuvanted pandemic influenza vaccine". Vaccine. 33 (46): 6173–6177. doi:10.1016/j.vaccine.2015.10.018. ISSN 1873-2518. PMID 26475444.
- ↑ 7.0 7.1 7.2 Carruthers, BM; van de Sande, MI; De Meirleir, KL; Klimas, NG; Broderick, G; Mitchell, T; Staines, D; Powles, ACP; Speight, N; Vallings, R; Bateman, L; Bell, DS; Carlo-Stella, N; Chia, J; Darragh, A; Gerken, A; Jo, D; Lewis, DP; Light, AR; Light, KC; Marshall-Gradisnik, S; McLaren-Howard, J; Mena, I; Miwa, K; Murovska, M; Stevens, SR (2012), Myalgic encephalomyelitis: Adult & Paediatric: International Consensus Primer for Medical Practitioners (PDF), ISBN 978-0-9739335-3-6
- ↑ Exley, Christopher; Swarbrick, Louise; Gherardi, Rhomain K.; Authier, Francois-Jérôme (February 1, 2009). "A role for the body burden of aluminium in vaccine-associated macrophagic myofasciitis and chronic fatigue syndrome". Medical Hypotheses. 72 (2): 135–139. doi:10.1016/j.mehy.2008.09.040. ISSN 0306-9877.
- ↑ Donegan, Katherine; Beau-Lejdstrom, Raphaelle; King, Bridget; Seabroke, Suzie; Thomson, Andrew; Bryan, Philip (October 9, 2013). "Bivalent human papillomavirus vaccine and the risk of fatigue syndromes in girls in the UK". Vaccine. 31 (43): 4961–4967. doi:10.1016/j.vaccine.2013.08.024. ISSN 0264-410X.
- ↑ 10.0 10.1 Liu, Xianfang C.; Bell, Christopher A.; Simmonds, Kimberley A.; Svenson, Lawrence W.; Russell, Margaret L. (April 4, 2016). "Adverse events following HPV vaccination, Alberta 2006-2014". Vaccine. 34 (15): 1800–1805. doi:10.1016/j.vaccine.2016.02.040. ISSN 1873-2518. PMID 26921782.
- ↑ Public Health Agency of Canada (January 18, 2012). "NACI Statement: HPV vaccine update". canada.ca. Retrieved September 30, 2021.
- ↑ Feiring, Berit; Laake, Ida; Bakken, Inger Johanne; Greve-Isdahl, Margrethe; Wyller, Vegard Bruun; Håberg, Siri E.; Magnus, Per; Trogstad, Lill (July 24, 2017). "HPV vaccination and risk of chronic fatigue syndrome/myalgic encephalomyelitis: A nationwide register-based study from Norway". Vaccine. 35 (33): 4203–4212. doi:10.1016/j.vaccine.2017.06.031. ISSN 0264-410X.
- ↑ Rodríguez-Galán, M. A.; Pérez-Vilar, S.; Díez-Domingo, J.; Tuells, J.; Gomar-Fayos, J.; Morales-Olivas, F.; Pastor-Villalba, E. (November 1, 2014). "Notificación de reacciones adversas a la vacuna frente al virus del papiloma humano en la Comunidad Valenciana (2007-2011)". Anales de Pediatría (in español). 81 (5): 303–309. doi:10.1016/j.anpedi.2013.11.031. ISSN 1695-4033.
- ↑ "HPV vaccine safety". National Health Service. July 31, 2019. Retrieved September 30, 2021.
- ↑ Arbyn, M.; Xu, L. (December 2, 2018). "Efficacy and safety of prophylactic HPV vaccines. A Cochrane review of randomized trials". Expert Review of Vaccines. 17 (12): 1085–1091. doi:10.1080/14760584.2018.1548282. ISSN 1476-0584. PMID 30495978.
- ↑ Peakman, Mark; Skowera, Ania; Hotopf, Matthew (2006), "Immunological dysfunction, vaccination and Gulf War illness", Immunological dysfunction, vaccination and Gulf War illness. Philosophical Transactions of the Royal Society B: Biological Sciences, 361 (1468): 681–687, doi:10.1098/rstb.2006.1826
- ↑ Bernton, E.; Hoover, D.; Galloway, R.; Popp, K. (December 29, 1995). "Adaptation to chronic stress in military trainees. Adrenal androgens, testosterone, glucocorticoids, IGF-1, and immune function". Annals of the New York Academy of Sciences. 774: 217–231. ISSN 0077-8923. PMID 8597461.
- ↑ Ramírez, F.; Fowell, D.J.; Puklavec, M.; Simmonds, S.; Mason, D. (April 1, 1996). "Glucocorticoids promote a TH2 cytokine response by CD4+ T cells in vitro". Journal of Immunology (Baltimore, Md.: 1950). 156 (7): 2406–2412. ISSN 0022-1767. PMID 8786298.
- ↑ Skowera, A.; Cleare, A.; Blair, D.; Bevis, L.; Wessely, S.C.; Peakman, M. (February 2004). "High levels of type 2 cytokine-producing cells in chronic fatigue syndrome". Clinical and Experimental Immunology. 135 (2): 294–302. doi:10.1111/j.1365-2249.2004.02354.x. ISSN 0009-9104. PMC 1808936. PMID 14738459.
- ↑ Patarca, Roberto (2001). "Cytokines and Chronic Fatigue Syndrome". Annals of the New York Academy of Sciences. 933 (1): 185–200. doi:10.1111/j.1749-6632.2001.tb05824.x. ISSN 1749-6632.
- ↑ "Immunocompromised". cancer.gov. February 2, 2011. Retrieved December 22, 2020.
- ↑ 22.0 22.1 22.2 Klimas, Nancy (March 13, 2020). "COVID-19 and ME/CFS - Dr Nancy Klimas, Mar 13, 2020". Retrieved March 24, 2020.
- ↑ Carruthers, Bruce M.; Jain, Anil Kumar; De Meirleir, Kenny L.; Peterson, Daniel L.; Klimas, Nancy G.; Lerner, A. Martin; Bested, Alison C.; Flor-Henry, Pierre; Joshi, Pradip; Powles, AC Peter; Sherkey, Jeffrey A.; van de Sande, Marjorie I. (2003), "Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Clinical Working Case Definition, Diagnostic and Treatment Protocols" (PDF), Journal of Chronic Fatigue Syndrome, 11 (2): 7–115, doi:10.1300/J092v11n01_02
- ↑ 24.0 24.1 Prinsen, Hetty; de Vries, I. Jolanda M.; Torensma, Ruurd; Pots, Jeanette M.; Mulder, Sasja F.; van Herpen, Carla M. L.; Elving, Lammy D.; Bleijenberg, Gijs; Stelma, Foekje F. (December 17, 2012). "Humoral and cellular immune responses after influenza vaccination in patients with chronic fatigue syndrome". BMC Immunology. 13 (1): 71. doi:10.1186/1471-2172-13-71. ISSN 1471-2172. PMC 3534525. PMID 23244635.
- ↑ 25.0 25.1 25.2 Arvas, Ahmet (September 1, 2014). "Vaccination in patients with immunosuppression". Turkish Archives of Pediatrics/Türk Pediatri Arşivi. 49 (3): 181–185. doi:10.5152/tpa.2014.2206. ISSN 1306-0015. PMC 4462293. PMID 26078660.
- ↑ 26.0 26.1 "Vaccination of Adults with Weakened Immune System". Centers for Disease Control and Prevention. November 12, 2020. Retrieved December 22, 2020.
- ↑ CDC (November 17, 2020). "Children and Flu Antiviral Drugs". Centers for Disease Control and Prevention. Retrieved December 22, 2020.
- ↑ Open Medicine Foundation (February 28, 2018). "OMF-funded research: T cells and molecular immunology". Open Medicine Foundation. Retrieved July 11, 2019.
- ↑ 29.0 29.1 29.2 29.3 29.4 29.5 National Institute of Allergy and Infectious Diseases. "Vaccine Types". niaid.nih.gov. Retrieved November 1, 2020.
- ↑ 30.0 30.1 30.2 30.3 30.4 Health and Human Services. "Vaccine Types". vaccines.gov. Retrieved November 1, 2020.
- ↑ "mRNA vaccines | COVID-19 and Your Health". Centers for Disease Control and Prevention. February 11, 2020. Retrieved December 22, 2020.
- ↑ Health and Human Services. "Vaccine Basics". vaccines.gov. Retrieved November 1, 2020.
- ↑ World Health Organization. "Coronavirus disease (COVID-19): Vaccines". World Health Organization. Retrieved November 1, 2020.
- ↑ Centers for Disease Control. "Vaccine Storage and Handling" (PDF). cdc.gov. Centers for Disease Control and Prevention.
- ↑ CDC (February 11, 2020). "Coronavirus Disease 2019 (COVID-19)". Centers for Disease Control and Prevention. Retrieved November 1, 2020.
- ↑ "Vaccines". blausen.com. Retrieved August 25, 2018.

