- This page was created by volunteers like you!
- Help us make it even better. To learn more about contributing to MEpedia, click here.
- Join the movement
- Visit #MEAction to find support or take action. Donate today to help us improve and expand this project.
Glycolysis
Glycolysis is the first step in a process known as cellular respiration. Glycolysis is the process of breaking down (-lysis) glucose (glyco-), a sugar molecule that provides energy for the human body. Glucose is a polymer (made up of a chain of carbons) and therefore it takes multiple steps to break it down into usable energy. There are a total of ten steps in glycolysis. After the final step, the products of the broken down glucose molecule will enter the next step of cellular respiration.
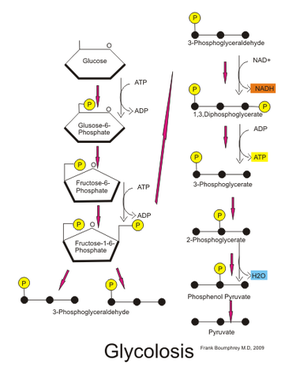
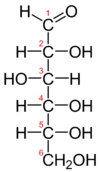
The differences between oxidative phosphorylation, anaerobic glycolysis, and aerobic glycolysis (Warburg effect) is detailed below:[1]
Oxidative phosphorylation
In the presence of oxygen, nonproliferating (differentiated) tissues first metabolize glucose to pyruvate via glycolysis and then completely oxidize most of that pyruvate in the mitochondria to CO2 during the process of oxidative phosphorylation. Because oxygen is required as the final electron acceptor to completely oxidize the glucose, oxygen is essential for this process.
Anaerobic glycolysis
When oxygen is limiting, cells can redirect the pyruvate generated by glycolysis away from mitochondrial oxidative phosphorylation by generating lactate (anaerobic glycolysis). This generation of lactate during anaerobic glycolysis allows glycolysis to continue (by cycling NADH back to NAD+), but results in minimal ATP production when compared with oxidative phosphorylation.
Aerobic glycolysis
Warburg observed that cancer cells tend to convert most glucose to lactate regardless of whether oxygen is present (aerobic glycolysis). This property is shared by normal proliferative tissues. Mitochondria remain functional and some oxidative phosphorylation continues in both cancer cells and normal proliferating cells. Nevertheless, aerobic glycolysis is less efficient than oxidative phosphorylation for generating ATP. In proliferating cells, approximately 10% of the glucose is diverted into biosynthetic pathways upstream of pyruvate production.
Glycolysis and myalgic encephalomyelitis
Abnormal levels of glycolysis metabolites have been observed in patients with myalgic encephalomyelitis (ME).[2] This suggests that individuals with ME may experience abnormal regulation of glycolysis within their cells.[3].
Another study also indicates impaired pyruvate dehydrogenase function in myalgic encephalopathy/chronic fatigue syndrome [4]

See also
Learn more
References
- ↑ van der Heiden, Matthew G.; Cantley, Lewis C.; Thompson, Craig B. (May 2009). "Understanding the Warburg Effect: The Metabolic Requirements of Cell Proliferation". Science.
- ↑ Nagy-Szakal, Dorottya; Barupal, Dinesh K.; Lee, Bohyun; Che, Xiaoyu; Williams, Brent L.; Kahn, Ellie J.R.; Ukaigwe, Joy E.; Bateman, Lucinda; Klimas, Nancy G. (July 3, 2018). "Insights into myalgic encephalomyelitis/chronic fatigue syndrome phenotypes through comprehensive metabolomics". Scientific Reports. 8 (1). doi:10.1038/s41598-018-28477-9. ISSN 2045-2322.
- ↑ Germain, Arnaud; Ruppert, David; Levine, Susan M.; Hanson, Maureen R. (2017). "Metabolic profiling of a myalgic encephalomyelitis/chronic fatigue syndrome discovery cohort reveals disturbances in fatty acid and lipid metabolism". Molecular BioSystems. 13 (2): 371–379. doi:10.1039/C6MB00600K. ISSN 1742-206X.
- ↑ Fluge, Øystein; Mella, Olav; Bruland, Ove; Risa, Kristin; Dyrstad, Sissel E.; Alme, Kine; Rekeland, Ingrid G.; Sapkota, Dipak; Røsland, Gro V.; Fosså, Alexander; Ktoridou-Valen, Irini; Lunde, Sigrid; Sørland, Kari; Lien, Katarina; Herder, Ingrid; Thürmer, Hanne; Gotaas, Merete E.; Baranowska, Katarzyna A.; Bohnen, Louis M.L.J.; Schäfer, Christoph; McCann, Adrian; Sommerfelt, Kristian; Helgeland, Lars; Ueland, Per M.; Dahl, Olav; Tronstad, Karl J. (2016), "Metabolic profiling indicates impaired pyruvate dehydrogenase function in myalgic encephalopathy/chronic fatigue syndrome", JCI Insight, 1 (21), doi:10.1172/jci.insight.89376
- ↑ "SparkNotes: Glycolysis". sparknotes.com. Retrieved October 24, 2018.
- ↑ "Overview of glycolysis". khanacademy.org.

